Magazine
Everything under the sun
The sun is essential for life, but overexposure harms people, animals and the materials we use to build our world. Protection comes in some surprising forms.
“The sun never repents of the good he does,” said Benjamin Franklin, one of the Founding Fathers of the USA. “Nor does he ever demand a recompense.” It rises every day and we think nothing of it, but without the sun our planet would be uninhabitable.
The sun, a giant nuclear reactor, provides us with the heat and light that are essential to all life on Earth. Its rays also help our bodies make vitamin D, which in turn helps us absorb calcium and phosphate from our diet – minerals that are important for healthy bones, skin and muscles. It boosts our immune systems and releases mood-enhancing serotonin. But too much exposure to the sun’s ultraviolet (UV) radiation is detrimental to everything on Earth – humans, animals, plants and objects. Dermatologists say there is no such thing as a “healthy” tan. Tanned skin is damaged skin.

Ultraviolet radiation from the sun reaches us in wavelengths between 100 and 400 nanometers (billionths of a meter). Around 5 percent of UV radiation is UVB, which only penetrates into the epidermis, while 95 percent is UVA. This can penetrate far deeper, though both UVA and UVB rays can cause skin cancer. And it’s not just humans that need protection – animals do too. In Cambodia, rats trained to sniff out deadly landmines need to have their ears covered in sunblock to allow them to carry out their life-saving work.
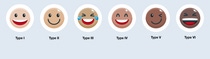
The six skin types
Developed in the 1970s by U.S. dermatologist Thomas Fitzpatrick, the Fitzpatrick scale remains the recognized guide to how likely skin is to suffer sun damage. It is even used as the basis for skin color in emojis.
Click on the plus sign to find more information about the different skin types.
Typ I
Haut: blassweiß, oft mit Sommersprossen, bekommt schnell einen Sonnenbrand und bräunt nicht
Augen: blau oder grün
Haar: blond oder rot
Typ I
Haut: blassweiß, oft mit Sommersprossen, bekommt schnell einen Sonnenbrand und bräunt nicht
Augen: blau oder grün
Haar: blond oder rot
Typ I
Haut: blassweiß, oft mit Sommersprossen, bekommt schnell einen Sonnenbrand und bräunt nicht
Augen: blau oder grün
Haar: blond oder rot
Typ I
Haut: blassweiß, oft mit Sommersprossen, bekommt schnell einen Sonnenbrand und bräunt nicht
Augen: blau oder grün
Haar: blond oder rot
Typ I
Haut: blassweiß, oft mit Sommersprossen, bekommt schnell einen Sonnenbrand und bräunt nicht
Augen: blau oder grün
Haar: blond oder rot
Type III
Skin: light brown, tans quickly, but still gets sunburn
Eyes: hazel or light brown
Hair: dark blonde or brown
The reason for reddening
General awareness about the risks of sun exposure is growing, but it was only in the last decade that researchers, led by Professor Richard Gallo, Chair of the Department of Dermatology at UC San Diego, USA, discovered why our skin reddens.
It is part of a process to alert the body that it is in danger. When skin cells are damaged by UV rays, it causes a type of ribonucleic acid (RNA) within those cells to break up. Our bodies become aware of the damage to the RNA because it is detected by what are called receptor molecules in neighboring cells. The receptor molecules instruct the body to inflame the skin around the damaged cell – which is what we experience as sunburn. “The reason for the inflammation is to clear away the sun-damaged cells,” says Gallo. “But it’s not only clearing, it’s actually alerting the body that the damage has occurred so you can have the appropriate response.”
As well as tests on human skin cells, the team carried out a study in mice and found that those without the receptor molecules did not have as much of a redness response. “At one point everything became predictable as a pathway,” says Gallo. Thanks to the research we now know that the receptor molecules are part of a process that warns us that our skin has been exposed to too much sunlight.

The right protection
While countries like Australia are doing a good job of getting sun safety messages across, others have a great deal of work to do, Gallo believes, with tanning still widely associated with exercise and outdoor activity. Cancer Research in the U.K., for example, has found a 150 percent rise in skin cancer deaths since the 1970s. “There’s this common misperception that tanning is healthy. But it’s very clear that there’s damage going on,” says Gallo.
Sunscreens to protect us from this damage have developed considerably since the first widely available product, developed by Austrian Franz Greiter, went on sale in 1946 with a sun protection factor (SPF) of just two. Getting the UV filters exactly right, however, can still be a difficult balancing act, as products need to protect against the different kinds of UV rays while still feeling pleasant on the skin.

There are two types of UV filter: organic filters based on carbon, and inorganic filters based on zinc and titanium oxide. Organic compounds absorb UV light while inorganic particulates not only absorb but reflect and scatter the light. One organic example is BASF’s Tinosorb® M. Around 90 percent of its action is via absorption and the rest through scattering. “We’ve chosen the most efficient molecules – an SPF 50 formulation absorbs 98 percent of the incoming UVB light, which is incredible with such a thin film,” says Marcel Schnyder, Head of the Global Technical Center Sun Care at BASF in Grenzach, Germany. Sunscreen products made with it have a light texture and dry feel, while meeting the UVA protection guidelines of the European Union.

These improvements, along with growing awareness of the dangers, have led to growth in the use of sunscreen products. But there is also a cost. A 2015 study estimated that up to 14,000 tons of sunscreen end up in the world’s oceans every year, potentially damaging coral reefs and marine life. This led BASF to launch the EcoSun Pass, which provides a comprehensive and transparent environmental evaluation of sun care products based on eight internationally recognized criteria including aquatic, terrestrial and sediment toxicity. “We created an algorithm that calculates the environmental impact of a sunscreen formulation, taking into account these eight parameters for each UV filter,” says Schnyder. The EcoSun Pass value for a given formulation is normalized for its sun protection factor. This is to prevent sunscreens being rated “poor” because of their high efficacy and filter concentration. The higher the EcoSun Pass value for a formulation, the more environmentally friendly it is. “Customers are given a comprehensive evaluation of their product’s filter systems, and manufacturers can develop sunscreens with the highest degree of environmental compatibility without compromising performance,” says Schnyder.
UV index
The UV index describes the daily peak value of the sunburn effective UV irradiance expected on the ground. Similar to the Richter scale for earthquakes, the UV index is to be understood as a scale that is open at the top and provides guide values for the UV irradiance. The higher the UV index, the higher the UV irradiance and the faster sunburn can occur on unprotected skin.
By clicking on the respective location in the world map, you can view the typical UVI values over the course of a year (maximum values, i.e. around noon, on the 21st of each month).
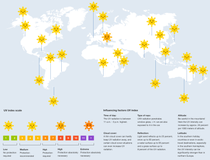
Berlin
Germany
Typical UV index values for the 21st of the respective month

Berlin
Germany
Typical UV index values for the 21st of the respective month

Berlin
Germany
Typical UV index values for the 21st of the respective month

Berlin
Germany
Typical UV index values for the 21st of the respective month

Berlin
Germany
Typical UV index values for the 21st of the respective month

Berlin
Germany
Typical UV index values for the 21st of the respective month

Berlin
Germany
Typical UV index values for the 21st of the respective month

Berlin
Germany
Typical UV index values for the 21st of the respective month

Berlin
Germany
Typical UV index values for the 21st of the respective month

Berlin
Germany
Typical UV index values for the 21st of the respective month

Berlin
Germany
Typical UV index values for the 21st of the respective month

Berlin
Germany
Typical UV index values for the 21st of the respective month

Berlin
Germany
Typical UV index values for the 21st of the respective month

Berlin
Germany
Typical UV index values for the 21st of the respective month

Berlin
Germany
Typical UV index values for the 21st of the respective month

Berlin
Germany
Typical UV index values for the 21st of the respective month

Berlin
Germany
Typical UV index values for the 21st of the respective month

Berlin
Germany
Typical UV index values for the 21st of the respective month

Berlin
Germany
Typical UV index values for the 21st of the respective month

Berlin
Germany
Typical UV index values for the 21st of the respective month

Berlin
Germany
Typical UV index values for the 21st of the respective month

Berlin
Germany
Typical UV index values for the 21st of the respective month

Berlin
Germany
Typical UV index values for the 21st of the respective month

Berlin
Germany
Typical UV index values for the 21st of the respective month

Crumbling and cracking
But it is not just our skin that needs protection. The sun’s radiation also affects everyday objects, weakening adhesives, causing colors to fade and reducing durability. Materials particularly at risk are polypropylene and polyethylene, which are found in a wide range of products, including plastic containers, clothing, cabling and machine parts. These things can crack or even disintegrate under the sun’s glare unless they are protected by light stabilizers.
“Bottle crates that stand in the sunlight begin to degrade in weeks. Then you can’t use them. But if you stabilize them you can use them again and again, for years,” says Daniel Müller, Head of Competence Center Film & Tape at BASF in Kaisten, Switzerland. “It’s the same with garden furniture. When it starts whitening, that is usually degradation of the polymer on the surface. With a stabilizer you can prevent this and prolong the lifetime.”
BASF produces two kinds of stabilizer – Ultraviolet Light Absorbers (UVA) and Hindered-Amine Light Stabilizers (HALS). HALS trap free radicals, which prevents surface cracking and helps maintain gloss. The best results are often achieved by combining the two. BASF’s Tinuvin® range comprises UV stabilizers for a variety of plastics, preventing color fading and other ageing signs. “With Tinuvin we provide protection and also repair of the polymer when it starts to degrade,” says Müller.

Like any outdoor paint, the sculptures are going to fade.”
Exposed to the elements
It is frustrating when garden furniture fades, but the stakes are far higher with valuable artworks. The damaging effects of UV rays are something museums have to contend with all the time. At the J. Paul Getty Museum in Los Angeles, USA, outdoor bronze sculptures are protected with wax at least annually, with lacquers often added on top of that.
One of the main challenges is protecting a piece without compromising it. Julie Wolfe is Associate Conservator at the museum. “Right now, I’m focusing on the artworks of Roy Lichtenstein, who made painted outdoor sculptures,” she says. “The paint he originally applied was something he created using industrial automotive and artist paints. Like any outdoor paint, they’re going to fade and need to be repainted. Now that he’s no longer here, we have to try to recreate his technique.”
Learning how thick the brush strokes should be, replicating the exact shade of color from a faded one – that takes a lot of time and research. “If he’d painted swatches and stored them in a dark, protected space, it would be perfect,” says Wolfe. “But that rarely happens with artists!”